 
In affected individuals lung function deteriorates progressively over several years, with increasing symptoms such as cough, sputum production, and dyspnoea. 1 COPD is associated with an enhanced chronic inflammatory response which is responsible for the airway abnormalities and architectural distortion of the lung parenchyma. 1ĬOPD is characterized by airflow limitation that is progressive and not fully reversible the latest severity categorization also includes exacerbation frequency and symptom burden as key features. 1 Globally the burden of disease is projected to increase in the coming decades due to continued exposure to COPD risk factors and an ageing population. It is currently the fourth leading cause of death worldwide and predicted to be the third by 2020. Anoro Ellipta Summary of Product Characteristics, May 2014.Chronic obstructive pulmonary disease (COPD) is a multi-component disease which is both preventable and treatable.Other commonly reported adverse effects included UTI, sinusitis, pharyngitis, upper respiratory tract infection, headache, cough, oropharyngeal pain, constipation and dry mouth. The most frequently reported adverse effect in patients receiving vilanterol/umeclidinium was nasopharyngitis. 1ĬOPD exacerbations were reduced by 50% with vilanterol/umeclidinium compared with placebo (HR 0.5, p=0.004), by 20% compared with umeclidinium (HR 0.8, p=0.391) and by 30% compared with vilanterol (HR 0.7, p=0.121).  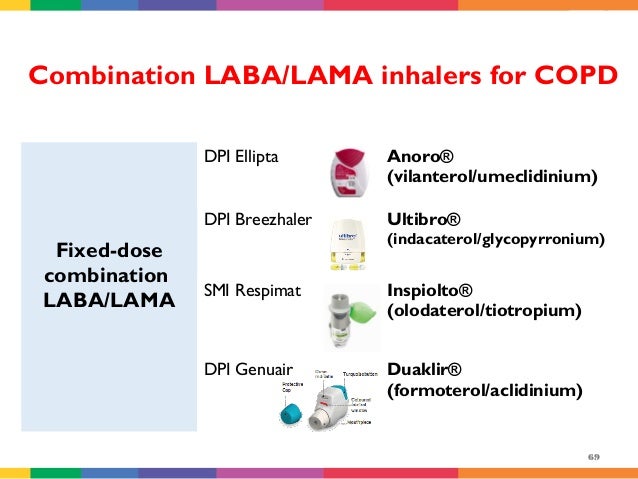
Similar outcomes were also observed with placebo and umeclidinium montherapy. Three six-month trials (total n=1747) showed that vilanterol/umeclidinium reduced the use of rescue medication compared with tiotropium. George’s Respiratory Questionnaire compared with tiotropium monotherapy. In one of the three comparator studies, the combination was associated with a statistically significant reduction in total score on the St. Improvements relating to health-related quality of life were also observed for vilanterol/umeclidinium in clinical studies. 1Īt week 24, a clinically meaningful increase in transition dyspnoea index (TDI) focal score (key secondary endpoint) was observed with vilanterol/umeclidinium compared with placebo (p<0.001). Vilanterol/umeclidinium was also shown to produce significantly greater improvements in lung function than tiotropium in two of three six-month active comparator studies (n=905, p<0.001 and n=410, p<0.001) and numerically greater improvements in the third (n=432, p=0.018). In one six-month study (n=1,532) significant improvements in lung function (as defined by change from baseline in trough FEV 1) at week 24 were observed for vilanterol/umeclidinium compared with placebo (p<0.001) and with each monotherapy treatment arm (p<0.001 vs vilanterol, p=0.004 vs umeclidinium). The clinical efficacy of once-daily vilanterol/umeclidinium was evaluated in eight phase III studies involving 6,835 adults with COPD. Its bronchodilatory action is complemented by that of umeclidinium, a long-acting muscarinic receptor antagonist (LAMA) which exerts its effects by competitively inhibiting the binding of acetylcholine to muscarinic receptors on airway smooth muscle. Vilanterol is a long-acting beta 2 agonist (LABA).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
